Description
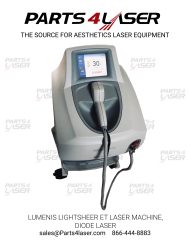
LUMENIS LIGHTSHEER ET
Multi-Application Capability
In addition to documentation of clinical efficacy with hair removal and permanent hair reduction, the LightSheer is also FDA cleared for the treatment of pseudo folliculitis barbae (PFB), benign pigmented lesions, and leg veins, providing the operator with multi-application capabilities and versatility.In addition to
– Leg Veins – Approximately 50% to 60% of men and women have unsightly leg veins. While sclerotherapy is still considered the treatment of choice for the removal of superficial leg veins, many vessels may be too small to inject or patients may have an aversion to this invasive procedure. The LightSheer diode laser system can provide an alternative method of treatment that requires no painful injections.
– Benign Pigmented Lesions – The LightSheer diode laser system can successfully treat a wide range of cosmetically undesirable benign pigmented lesions.
– Pseudofolliculitis Barbae (PFB) – PFB, or razor bumps, is a common condition in which the regrowing hair, after shaving, curves back towards and pierces the skin resulting in a pustule or papule. PFB is very prevalent in populations with curly hair, affecting nearly 85% of men of African descent and a growing number of women. The LightSheer’s excellent safety profile for all skin types including the darkest skin, makes it a safe, effective and revolutionary alternative for the treatment of pseudofolliculitis barbae.
The Right Choice for Optimal Safety and Efficacy
The LightSheer system was designed from the ground up for effective hair removal, providing unsurpassed safety and efficacy, reliability, and ease-of-use. Its advanced high-power diode technology delivers high fluence combined with a large spot size, user-selected pulse width, aggressive contact cooling and compression for high-performance, unmatched by other high-tech hair removal systems. Both in rigorous scientific trials as well as extensive clinical practice, LightSheer system has emerged as the gold standard in laser hair removal.
Parts4laser is not affiliated, associated, authorized, endorsed by, or in any way officially connected with Lumenis their affiliates. Any logos and trademarks of third parties that may be found on our website are registered trademarks of their respective owners. The use of any trademark is for identification and reference purposes only and does not imply any association with the trademark holder.